Leadership quality is a decisive factor in venture capital (VC) success; investors should scrutinize founder and executive capabilities as rigorously as they evaluate scientific innovation. In our recent study on leadership in VC-backed companies (WittKieffer, 2025), we found that organizations that proactively assess, develop, and evolve their leadership teams across critical growth stages dramatically outperform those that treat talent as an afterthought. The adage in biotech investing — "bet on the jockeys, not the horse" — has never been more relevant: even the most promising science can falter without exceptional leadership capable of pivoting when data shifts, maintaining stakeholder alignment through setbacks, and executing against milestones with precision.
"The quality of the leadership team is so important because, if the science doesn't deliver, a very good team will find a way to rebound. A mediocre team with great science will eventually fail, but a great team with mediocre science will pivot and succeed."
But how do biotech companies operationalize this insight? Their challenge is particularly acute, as biotechs face multiple critical junctures that require different organizational capabilities to meet the next milestones. The leadership capabilities required to validate a scientific hypothesis in pre-clinical and Phase I, for example, bear little resemblance to those needed to prepare for commercial launch in Phase III, spanning clinical execution, regulatory navigation, and market readiness. Yet leadership transitions remain one of the most poorly managed aspects of biotech company building, frequently handled reactively rather than proactively.
"You're writing a check to a person, not to an idea. If the team can't execute or doesn't work well together, it's a no-go. Leadership quality is our top investment priority."
To address this challenge, we introduce the concept of milestone leadership, a structured approach that encompasses intentionally building organizational capabilities, leadership team composition, and culture in lockstep with a company's development milestones. Drawing on our work with biotech investors, CEOs, and board members, this framework provides a practical roadmap for anticipating leadership needs before value creation stalls, supporting smarter transitions at critical junctures, and maximizing upside as companies scale.
The Leadership Scorecard: Defining Success at Each Milestone
Before exploring how leadership must evolve across biotech milestones, we need to first establish what success looks like at each stage. This is where the leadership scorecard becomes essential: a structured framework that defines the specific capabilities, behaviors, and outcomes required for leadership effectiveness in a specific role at a given stage of company development.
In our work with venture-backed companies, we observe that the most successful investors and boards introduce scorecards early, ideally at the seed stage, to create shared clarity between founders, boards, and investors about leadership expectations. The scorecard serves multiple functions: it provides clarity on what a role is expected to deliver, establishes an objective basis for evaluating current organizational capabilities, and identifies gaps that must be addressed through complementary hires or development while mitigating selection risk.
Critically, the scorecard is not static. A leader who merits an A+ rating in pre-clinical stages may become a liability in later stages without developing new capabilities or being complemented by leaders who possess them. A founder-CEO who excels at articulating scientific vision and managing capital planning (essential scorecard criteria for Phase I) must later demonstrate operational discipline, regulatory navigation, and commercial strategy (Phase II-III scorecard criteria) or risk becoming a bottleneck to growth.
The power of the scorecard lies in its forward-looking application: it enables systematic assessment of leadership capabilities against evolving requirements. By regularly evaluating leaders against both present and anticipated scorecard criteria, companies can identify capability gaps 12-18 months before they become critical. This approach shifts leadership transitions from reactive crisis management to proactive strategic planning. Companies can coach their leadership team, make strategic hires, and manage succession decisions with objectivity and minimal disruption. Ultimately, the scorecard transforms leadership planning from subjective judgment into a measurable strategic asset.
WittKieffer's Milestone Leadership Framework
Our Milestone Leadership framework recognizes that what works in pre-clinical discovery rarely suffices for commercial scale. The framework is built on four interconnected dimensions that must evolve in concert as companies progress from scientific discovery to commercial readiness.
Capabilities ("Who"): Identifying the specific organizational capabilities required at each development stage. This dimension addresses how capabilities must shift as companies move through scientific discovery, scientific validation, clinical validation, and commercial stages, explicitly navigating the often-difficult transition from founding teams to professional management.
Timing ("When"): Determining the optimal moment to bring in specific functional expertise at the C-suite and senior leadership levels. Strategic timing involves proactive planning 12–18 months in advance of major milestones to institutionalize functional needs, scale services, and professionalize teams before execution gaps arise — rather than reacting when functional needs first emerge.
Culture ("How"): Managing the deliberate evolution of organizational identity by preserving the innovation DNA (urgency, innovation, and risk-taking) that characterizes early-stage biotechs while defining cultural non-negotiables and building the governance and quality systems required for late-stage development and commercial readiness.
Alignment ("Why"): Ensuring the leadership team functions as a unified body with collective accountability for enterprise-level goals by establishing clear decision-making cadences that create a multiplier effect, balancing individual functional excellence with C-suite cohesiveness, and creating systems for continuous improvement as organizations scale.
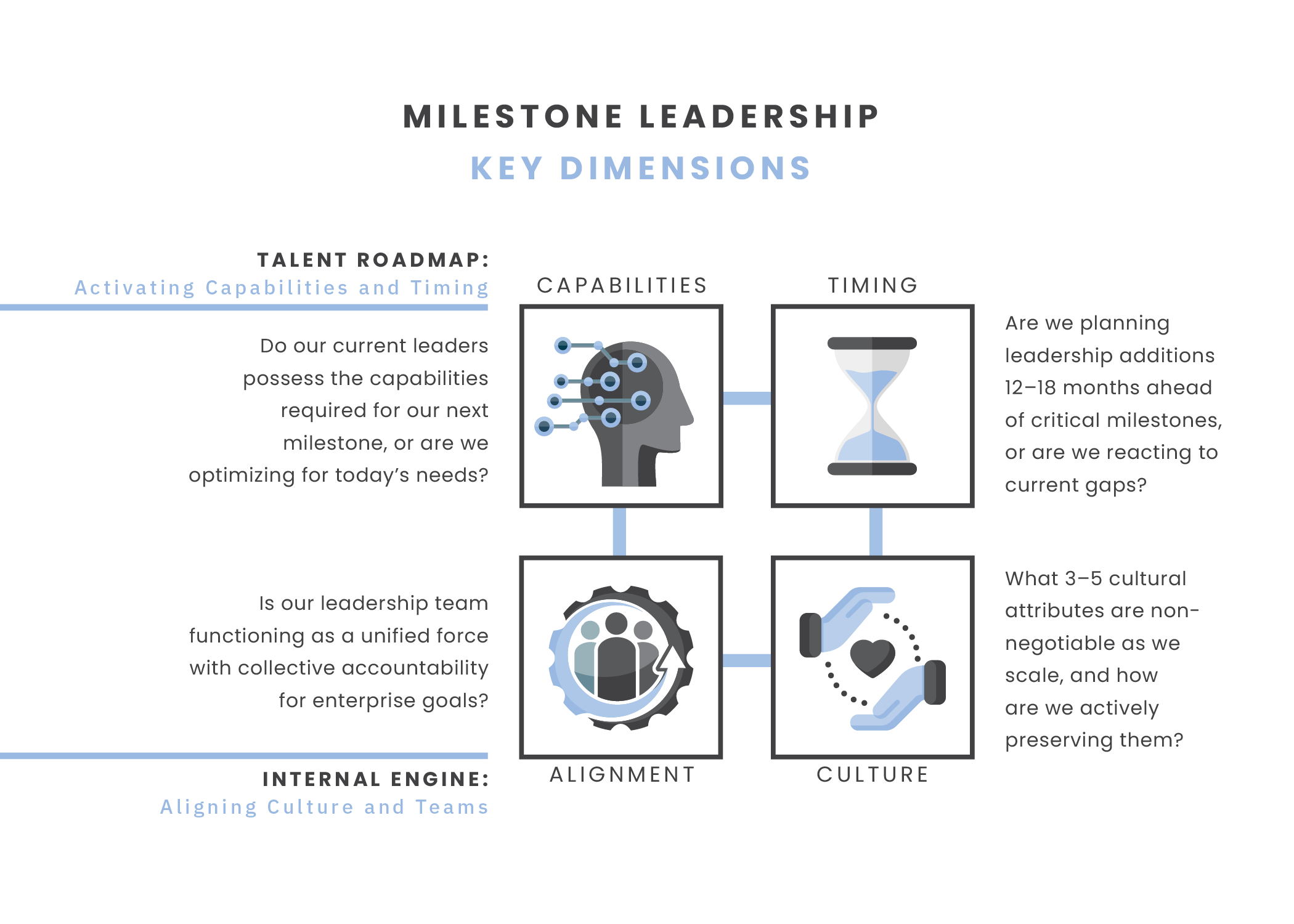
These four dimensions operate interdependently, not in isolation. Capability requirements drive timing decisions; timing influences culture evolution; and alignment practices reinforce both capability development and cultural integrity. Together, they create an organizational blueprint that must be assessed at each critical juncture, ensuring that as science advances toward real-world impact, leadership evolves in parallel.
"What is your priority? Have you just achieved a milestone, or is a milestone coming up? Are you in an execution phase, a fundraising phase, or a partnership phase? Once I've determined what stage the science is at, I can decide whether we need scientists or operational people who can execute."
The Talent Roadmap: Activating Capabilities and Timing
Biotech companies progress through particularly distinct development stages, each demanding fundamentally different organizational capabilities. Our Milestone Leadership Framework maps these transitions, showing how the leadership scorecard must be reconsidered at each critical juncture to reflect new priorities, leadership requirements, and success metrics.
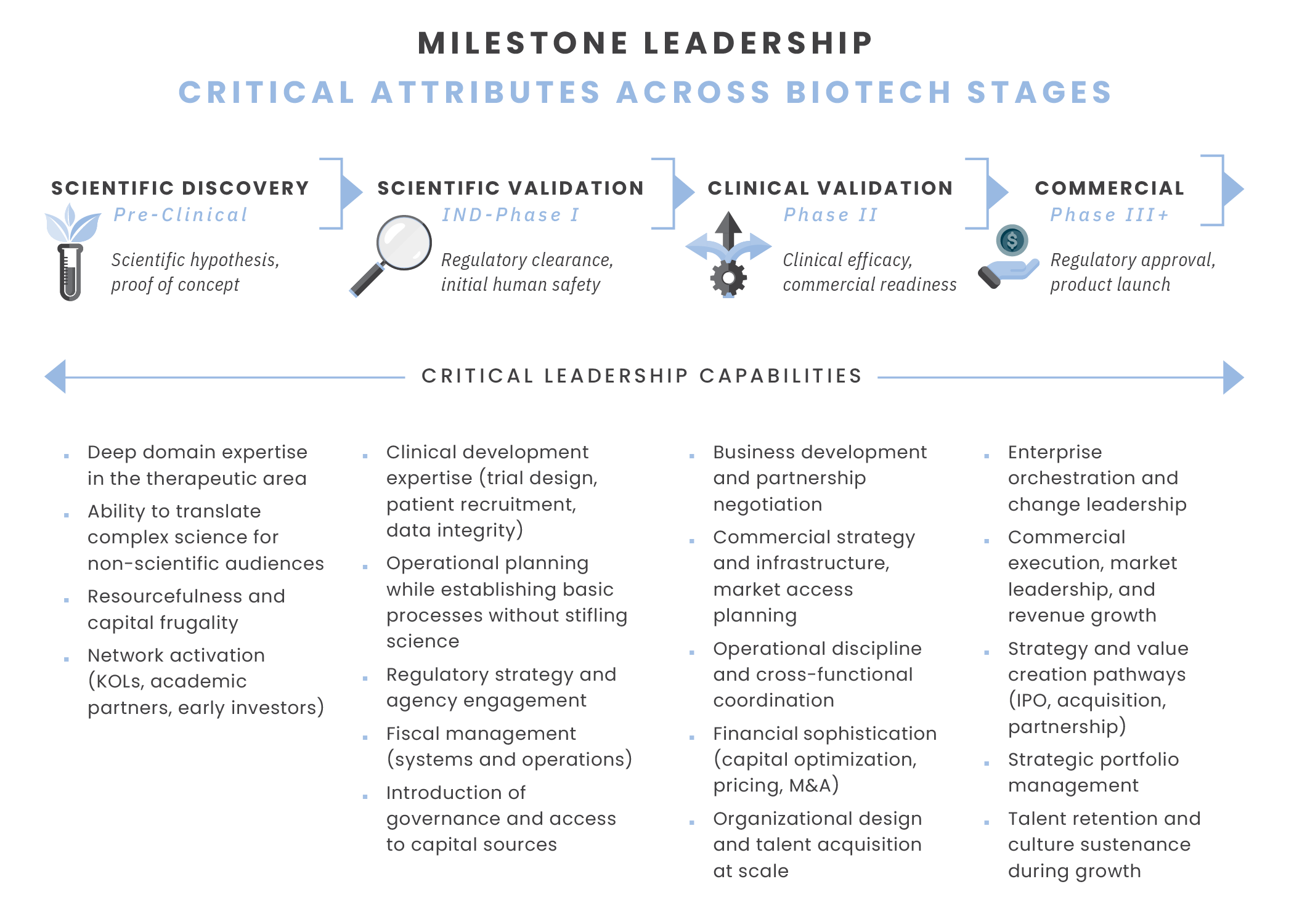
Stage 1: Scientific Discovery (Pre-Clinical)
Primary milestone: Validate the scientific hypothesis and establish proof of concept through in vitro and in vivo studies that assess safety, toxicity, and efficacy of a drug candidate before testing in humans.
Critical organizational capabilities: At this foundational stage, scientific credibility and cash preservation are prioritized. Success is measured by the ability to articulate a compelling biological rationale that links the scientific strategy to a viable market need, to translate complex science for non-scientific audiences (investors, board members, advisors), and to manage cash runway with discipline. Organizations maintain lean teams, often consisting of just two key roles, such as CEO and CSO, as their core leadership. Leaders are hands-on players, essential to executing key tasks — not just directing the work of others.
Successful leadership teams distinguish themselves through deep domain expertise that commands credibility with scientific advisors and potential academic partners, combined with the ability to activate external networks. This networking, meaning cementing relationships with KOLs, academic partners, and early-stage investors, becomes essential for building momentum and validating the scientific approach.
Stage 2: Scientific Validation (IND–Phase I)
Primary milestone: Achieve regulatory clearance and demonstrate initial human safety and dosage. Phase I typically takes several months, with approximately 70% of drugs advancing to the next phase (U.S. Food & Drug Administration, 2018).
Critical organizational capabilities: As companies enter the clinic, organizational needs shift to emphasize agility and regulatory navigation. New capabilities become critical to advancing the science from laboratory to human trials.
Clinical development expertise now becomes critical, encompassing trial design and management, patient recruitment strategies, and data integrity management. Leadership must demonstrate operational planning capability while establishing basic processes that bring structure without stifling the scientific innovation that fueled early success. This delicate balance — professionalizing operations while preserving entrepreneurial agility — often determines whether companies successfully navigate the IND filing and Phase I execution.
Regulatory strategy and agency engagement are non-negotiable at this stage. Leadership teams must build credible relationships with regulatory authorities, anticipate potential concerns, and design development pathways that satisfy both scientific rigor and regulatory requirements. Equally critical is early commercial scoping (i.e., defining total addressable market, differentiation strategy, and payer pathways), which companies often accomplish through interim commercial leaders, consultants, or specialized agencies to inform trial design and establish a clear path to market. In today's constrained capital markets, this commercial intelligence strengthens fundraising narratives and gives companies a competitive advantage at launch.
Fiscal management emerges as a critical capability, requiring leadership to implement more robust systems to manage the increased complexity of clinical budgets, resource allocation, and cash flow planning. The introduction of governance structures and strategic access to capital becomes essential as companies start raising seed or Series A fundraising rounds, prepare to bring in new investors in the next stage, and establish board-level oversight. Leadership's ability to present a compelling investment case and successfully attract and secure capital becomes a key scorecard metric.
Companies that fail to evolve in conjunction with the scorecard requirements often experience fundraising barriers and erosion in board and investor confidence, even when the underlying science remains sound.
Stage 3: Clinical Validation (Phase II)
Primary milestone: Demonstrate clinical efficacy and side effects and prepare for commercial readiness, partnership, or exit. This stage typically takes from several months to two years, with approximately 33% of drugs advancing to the next phase (U.S. Food & Drug Administration, 2018).
Critical organizational capabilities: This stage represents the most dramatic scorecard shift, expanding criteria to include commercial integration and strategic positioning. Investors now assess whether leadership can transition from "proving the science works" to "proving the business works."
The evolved scorecard becomes exponentially complex, consistent with the diversity of capabilities required to satisfy business, market, investor, regulator, payer, provider, and patient needs. Business development capabilities are a first priority, followed by market access and commercial strategy and infrastructure. Leadership teams must articulate competitive positioning, pricing strategy, and reimbursement pathways alongside therapeutic differentiation. Operational discipline and cross-functional coordination now become formalized to scale. Financial sophistication around capital allocation and optimization, pricing, and M&A distinguishes leadership teams who can create strategic paths from those who remain tactically focused. Organizational design and talent acquisition at scale test leadership's ability to build teams that can execute on increasingly complex mandates while evolving the culture that attracted early talent.
This is where CEO transitions most frequently occur, representing arguably the most critical inflection point in a biotech company's leadership evolution. At this juncture, more than at any other, boards and investors face a critical decision: can a founder-CEO who commanded credibility in early scientific and development stages evolve into a commercial leader capable of proving the business works and securing the funding and/or partners required to advance through subsequent stages? Or, must the company bring in a professional CEO with commercial expertise and capital markets fluency? Many companies struggle most acutely with this capability, making it a decisive factor in the transition decision.
Stage 4: Commercial (Phase III+)
Primary milestone: Achieve regulatory approval while efficacy testing and monitoring of adverse reactions continue, launch product, and scale revenue. This stage typically takes from one to four years, with approximately 25-30% of drugs advancing to the next phase (U.S. Food & Drug Administration, 2018).
Critical organizational capabilities: The capability shift here prioritizes enterprise orchestration and strategic value creation. Leaders must now balance multiple constituencies — patients, providers, payers, regulators, employees, and investors — while evolving the entrepreneurial culture that drove early success.
Commercial execution, market leadership, and revenue growth become the primary measures of effectiveness. Leadership must demonstrate market traction, optimize pricing and reimbursement strategies, and scale sales operations to capture market opportunity. At this point, most companies are already public, having raised the substantial capital required to reach commercialization. Value creation now centers on meeting or exceeding Wall Street analyst and investor expectations — a fundamentally different dynamic than earlier stages. The shift from pre-commercial to commercial public company requires distinct capabilities: managing quarterly earnings pressures, maintaining analyst relationships, balancing short-term performance with long-term pipeline development, and navigating the heightened scrutiny of public markets.
Strategic portfolio management becomes essential as companies look beyond the initial product to pipeline expansion through lifecycle management, label expansion, and external innovation partnerships. Compliance and operational readiness emerge as critical capabilities, requiring leadership teams to establish enterprise-grade systems for regulatory compliance, quality management, and commercial operations as the company transitions into late clinical and commercial stages.
Talent retention and culture sustenance during complexity and growth test leadership's ability to evolve the innovation DNA and entrepreneurial spirit that fueled early success while professionalizing operations to meet the demands of a scaled enterprise. The most successful companies retain the essential attributes of prior stages, such as scientific rigor and execution discipline, adding new capabilities without abandoning their roots.
Effective leadership transitions require proactive planning, transparent communication, and dedicated support for both incoming and outgoing leaders. From an investor perspective, a thoughtful evolution of the C-suite, aligned specifically with the company's development roadmap, serves as a powerful signal of organizational maturity and a primary indicator of readiness for the next stage of value creation.
Putting Milestone Leadership into Practice
"Talent attracts talent. The caliber of your early hires sets the tone for the intellectual quality of the company and the work it produces."
To ensure leadership keeps pace with scientific and clinical progress, boards and investors should build and adopt upfront a talent roadmap that emphasizes intentional leadership team construction supported by leadership assessment, structured team alignment, and ongoing development. Having a clear understanding of the leadership transitions needed, ideally before investing, enables biotech companies to build leadership capacity ahead of critical milestones rather than scrambling reactively when gaps emerge.
Role evolution. As companies progress from scientific discovery to commercial scale, the capabilities required of key leadership roles must evolve accordingly. There are distinct patterns in how key leadership roles evolve across the biotech lifecycle to drive success at each critical milestone.
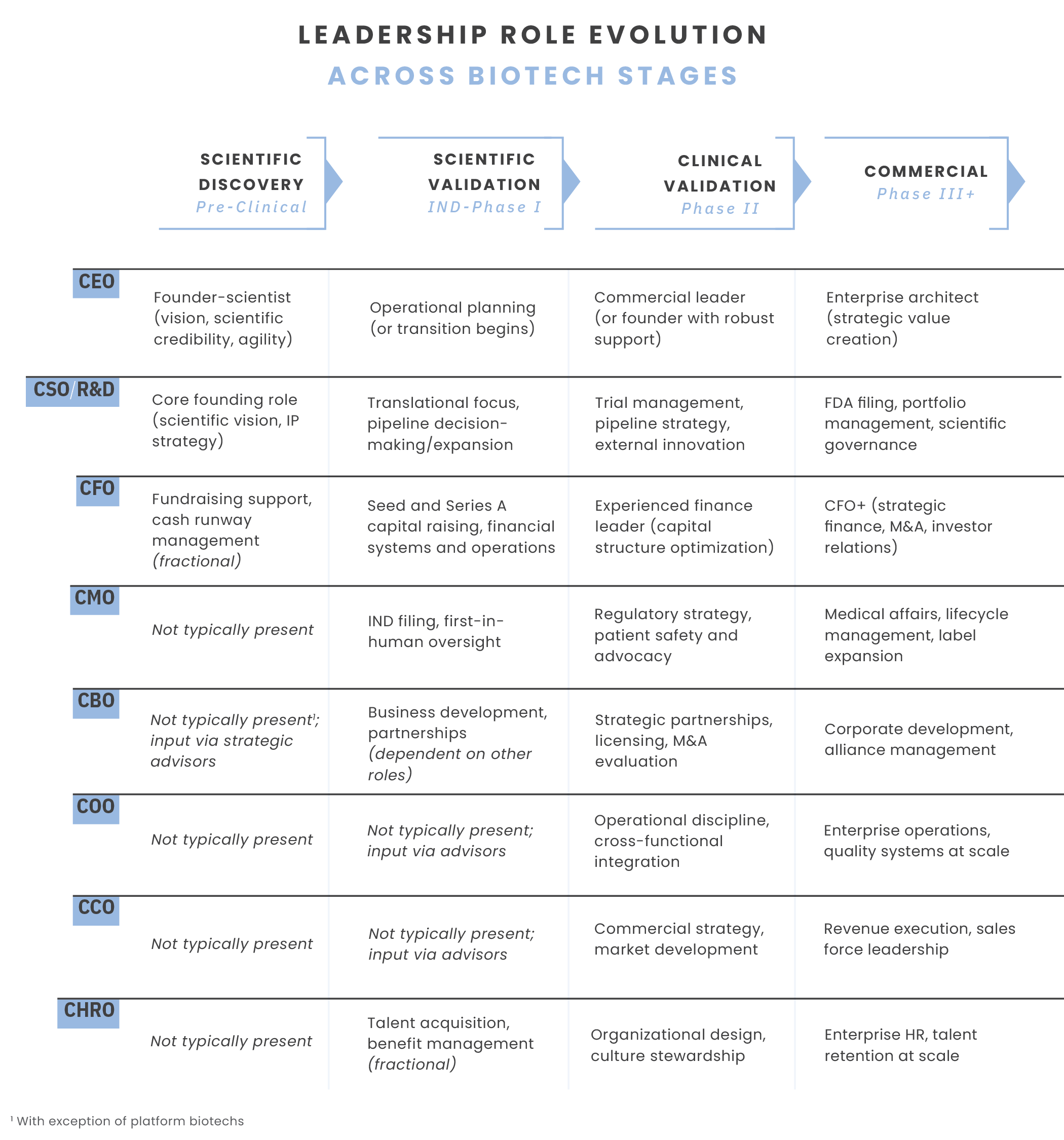
Strategic timing. Initiate leadership assessment and transition planning 12–18 months in advance of major milestones. Use this window to validate the specific capabilities required for the next stage rather than solving for current gaps. This forward-looking approach prevents the reactive rushing that often derails momentum at critical junctures.
"We look for the triple H: head, heart, and hunger. Intellectual and astute, passionate about the mission, and humble — eager to learn and eager to contribute."
The Internal Engine: Aligning Culture and Teams
While capabilities and timing define the "who" and "when," culture and team alignment define the "how" and "why", transforming talented individuals into a unified leadership body capable of navigating the biotech industry's unique paradoxes. By investing in intentional culture shaping and team alignment, companies ensure their leadership is more than a collection of ambitious individuals. Instead, it becomes a synchronized force that guides the organization through complex transitions and toward ambitious milestones. In an industry where scientific breakthroughs and leadership excellence must advance in tandem, team alignment and culture are the internal engine that powers sustainable value creation.
"Before we went into the clinic, I invested heavily in team dynamics to alleviate potential problems down the line. I wanted to prevent problems arising rather than dealing with them after they occurred."
Culture shapes the behaviors that either accelerate or impede execution. In biotech, the primary challenge is a cultural paradox: preserving the entrepreneurial spirit that fueled initial breakthroughs while building the structured processes, governance, and infrastructure necessary for late-stage development and commercial readiness. To achieve this, culture must be intentionally defined, managed, and evolved over time — not by abandoning founding principles, but by adapting behaviors and norms to new realities while preserving essential values. The driving elements of early-stage biotech culture, such as innovation, science as mission, calculated risk-taking, and bias toward urgency, are often at risk of dilution as organizations grow.
Impactful biotech leaders establish clear cultural non-negotiables — i.e., core values that remain inviolable regardless of growth stage, organizational size, or geographic expansion. By defining these elements explicitly, companies create guardrails that guide hiring, decision-making, and organizational design as they scale.
"The time to build cohesion is well before growth and transition — not during. High performance requires trust, and trust comes from working on projects together, connecting personally outside work, and conducting after-action reviews."
The executive team serves as both architect and model of culture through:
- Proactive culture management: Developing mechanisms to consistently monitor cultural health, identifying drift before it becomes entrenched.
- Trust-building leadership: Aligning stated values with demonstrated behaviors to create the psychological safety necessary for innovation.
- Collaborative culture shaping: Partnering (rather than delegating to HR) to consciously shape an evolved culture that supports the next growth stage.
Milestone leadership requires a deliberate approach that expands the organization's culture story, signals, and symbols; makes culture a recurring leadership focus; and celebrates along the way by reinforcing values and acknowledging exemplars. This enables biotech companies to enact cultural change with agility without sacrificing their organizational identity.
"Companies go through different stages of life, and the internal personality changes as they grow — not necessarily a bad thing. Rather, it’s important to acknowledge and manage this evolution while trying to preserve the parts of the company that you really want to maintain from the early stages."
Executive team alignment creates a multiplier effect where the team's combined impact exceeds the sum of individual contributions. Three pillars sustain alignment:
"We have an enterprise mindset and a no-jerks policy. Check ego and ambition at the door, show up as your authentic self, be vulnerable enough to admit mistakes, deal with ambiguity, and shift gears with agility."
- Collective priorities: Clarity on what the team collectively owns, above and beyond each executive's functional role, is essential. In biotech, where resource allocation significantly impacts research pipelines and market positioning, this prevents executives from pursuing departmental goals at the expense of enterprise objectives.
- Team norms and relationships: Defined norms and strong relationships foster the inclusive environment needed for innovation and paradox management. Biotech leaders must navigate inherent tensions: risk-taking versus prudence, speed versus thoroughness, scientific exploration versus commercial viability. Aligned teams develop the psychological safety to address these paradoxes openly.
- Effective decision-making: Aligned teams make more nuanced decisions and execute more effectively, enabling faster responses to market opportunities, regulatory challenges, and competitive threats — all critical in an industry where timing determines success.
Balancing individual accountability with collective C-suite performance requires two complementary approaches. First, evaluating executives on both functional metrics and contributions to leadership team effectiveness to ensure all executives focus on both departmental excellence and organizational success. Second, establishing mechanisms for reflection, feedback, and growth, including leadership retreats, 360-degree feedback, team coaching, or structured debriefs, to institutionalize continuous improvement as the team evolves alongside company growth.
The Milestone Leadership Advantage
The biotech journey from scientific innovation to commercial success requires more than groundbreaking science. It demands leadership that evolves deliberately with each milestone. As explored throughout this report, effective milestone leadership operates across four critical dimensions: advancing capabilities to match development stages, timing transitions proactively, preserving cultural strengths during scaling, and creating alignment across the leadership team.
By embedding a leadership scorecard and deploying a talent roadmap, biotech companies transform leadership evolution from a reactive hurdle into a strategic advantage — one that investors recognize, reward, and increasingly demand. Biotech boards and leadership teams that master these dimensions gain a significant competitive advantage: they anticipate leadership needs 12–18 months ahead of critical milestones, balance scientific expertise with commercial acumen, implement systems that evaluate both individual and collective performance, and actively manage cultural evolution.
In an industry where scientific breakthroughs and leadership excellence must advance in tandem, the Milestone Leadership Framework provides a roadmap for sustainable growth and value creation.
Your next milestone is approaching. Is your leadership ready?
Acknowledgments
This report represents a collaborative effort across WittKieffer's Global Life Sciences market. We extend our appreciation to Ayesha Price, Evan Markou, Jennifer Braverman, Luigi Frezza, Robert Nickey, Saule Serikova, and Ysette Witteveen for their insights, expertise, and dedication to advancing leadership excellence in the biotech industry.
We are equally grateful to the industry leaders who generously shared their experiences and perspectives through in-depth interviews. Their candid insights into the challenges and opportunities of biotech leadership transitions enriched our understanding of milestone leadership and informed the practical guidance presented throughout this report.
Key contributors to this research include:
Abigail Jenkins, President, Chief Executive Officer, and Board Member at a stealth-mode biotech company; Board Director at XORTX Therapeutics and Aquestive Therapeutics
Alicia Tozier, former Chief Strategy Officer at UNITY Biotechnology; former Commercial Leader at Genentech; currently Commercial and Strategy Consultant to biotech companies
Alison Finger, Chief Operating Officer at Vicero Bio; Board Director at KeifeRx and ADMA Biologics
Atul Saran, Chief Executive Officer at IntegerBio; Board Director at Proniras Corporation
Filippo Petti, Chief Executive Officer at Leucid Bio
Gregory Mayes, President and Chief Executive Officer at Reunion Neuroscience
Helen McBride, PhD, Partner at BOLD Capital Partners
Hugo Fry, Chief Executive Officer at Topas Therapeutics
James Sapirstein, former Chief Executive Officer at Alliqua, Tobira Therapeutics, and Contravir Pharmaceuticals; Board Director at Biotechnology Innovation Organization (BIO), sitting on the Emerging Companies Section and Health Section Boards
Laura Brand, Commercial Advisor to biotech companies; former Commercial Executive at Amgen and Celanese; former Consultant at Boston Consulting Group
Marc de Garidel, Chief Executive Officer at Abivax; Board Chair at Ipsen; Board Director at Claris Bio
Richard Christie, MD, PhD, Advisor to biotech and health tech companies
Robert Klein, PhD, Founder and Chief Executive Officer at Karnateq
Seg Reille, Commercial Strategy and Alliance Advisor to biotech companies; former Commercial Operations and Alliance Management Executive at Amgen; former Consultant at Bain & Company
Shetal Vyas, Senior Vice President, Commercial Strategy & Development at Ardelyx; Board Director at KJT
Stella Vnook, PhD, Chief Executive Officer at Likarda; Founder and Board Chair at Oral Biolife; Co-Founder and Board Director at Kaida BioPharma; Board Director at Firefly Neuroscience
Teymour Boutros-Ghali, PhD, Managing Partner at BOLD Capital Partners








